

News
10 Mar 2024
Press Release
5 Nov 2018 at 08:08
Bonn, 05.11.2018. Only few processes are more complex than the interplay of neurons within the brain. The intertwined circuitry, responsible for the behavior of organisms, is often difficult to identify. It's also hard to differentiate the circuits from other functional cell populations. For some years now, it has been evident that the most efficient way to examine the function of neural cells is the targeted activation or deactivation of cells. For this purpose, genetic ŌĆśtoolsŌĆÖ have been invented that allow cell activation by using a ray of light. Just recently, a group of researchers, among them Dr. Reinhard Seifert (caesar), finally succeeded in also selectively deactivating a single neuron with the help of light. This tool primarily consists of two subunits: a photoreceptor, which absorbs blue light and thereupon emits a messenger substance, and an ion channel, which reacts to the latter by opening up. The crucial factor is that the ion channel transports potassium which has an inhibiting effect on neurons ŌĆō a mechanism also observable in natural cell control. Thereby, it was made possible to control a broad spectrum of different neurons.
The shut-off impulse is induced by a sudden flash of blue light directly aimed at the tissue. This puts the cells into a state of rest for several minutes. Here, the receptor's sensitivity to light is remarkable. Very few photons trigger the effect. With that, it is possible to selectively control cells and to reduce side effects. The reversibility of that operation is also of great importance. After a short amount of time, the neurons will revert into their normal state without any lasting effects. Apart from neurons, the researchers also succeeded in integrating the tool into heart cells.
In the field of researching neural circuits the targeted activation and deactivation of single cells is a big advantage. It allows identifying the role of every single cell in the great population of neurons and the brain itself. Subsequently, it also illuminates the effect that a loss of single neurons has on the behavior.
The research work was carried out with the participation of researchers from the Humboldt-Universit├żt of Berlin, the German Center for Neurodegenerative Diseases in Berlin, the Charit├® Universit├żtsmedizin Berlin, the University of Bonn, caesar (Center of advanced European studies and research), the University of Freiburg and the Max Planck Institute for Neurobiology in Martinsried.
Publication:
Bernal Sierra, Yinth Andrea, Benjamin R. Rost, Martin Pofahl, Ant├│nio Miguel Fernandes, Ramona A. Kopton, Sylvain Moser, Dominik Holtkamp, Nicola Masala, Prateep Beed, John J. Tukker, Silvia Oldani, Wolfgang B├Čnigk, Peter Kohl, Herwig Baier, Franziska Schneider-Warme, Peter Hegemann, Heinz Beck, Reinhard Seifert, and Dietmar Schmitz. "Potassium Channel-Based Optogenetic Silencing." Nat Commun 9, no. 1 (2018/11/05 2018): 4611. http://dx.doi.org/10.1038/s41467-018-07038-8.
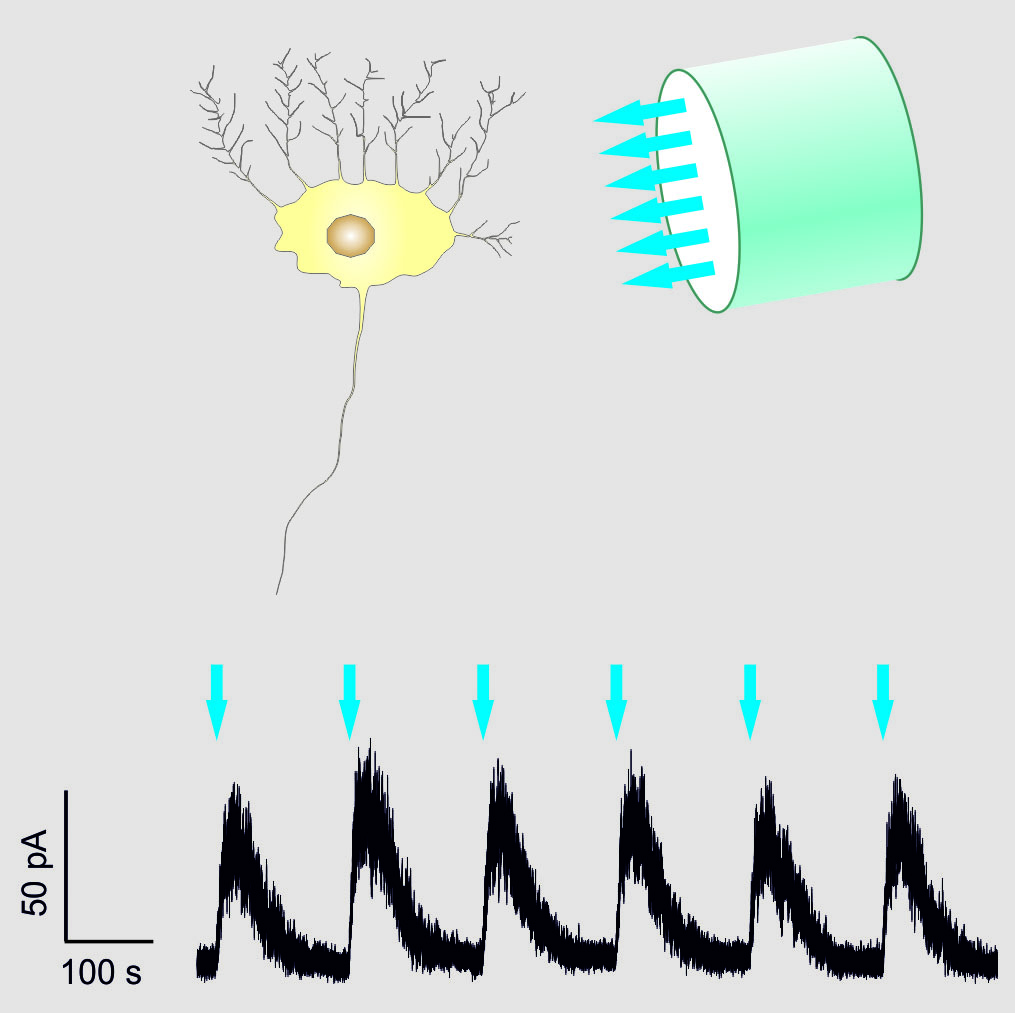
Figure: The photoreceptor was placed within a cell to selectively deactivate the neural activity with a ray of light. (Picture: R. Seifert / caesar)
Scientific contact: Dr. Reinhard Seifert (eMail: Reinhard.Seifert@caesar.de)
Press contact: Sebastian Scherrer (Tel.: +49 228/9656-139 / eMail: sebastian.scherrer@caesar.de)
About research center caesar
The Center of Advanced European Studies and Research (caesar) in Bonn, Germany, is a neuroethology research institute that focuses on the neural mechanisms underlying animal behavior. The institute is operated by a non-profit foundation under private law. Trustors are the Federal Republic of Germany and the Federal State of North Rhine-Westphalia. caesar is closely associated with the Max Planck Society (MPG) and shares its scientific principles. The President of the MPG chairs the Board of Trustees and caesar's directors are Scientific Members of the MPG.