Research News
Sperm cells are unique. Compared to sensory, heart, or nerve cells, sperm often use special proteins that do not exist in other cell types. Using various physical and chemical signposts, sperm navigate to the egg. Navigation is controlled by biochemical signalling pathways in sperm. Only in recent years has it been recognized that molecules familiar to us from somatic cells - such as olfactory cells or photoreceptors - do not play a role in sperm cells. Even more puzzling, the signal molecules in sperm look like familiar faces on the outside - that is, their amino-acid sequence is similar to that of their cousins in somatic cells. However, it is a "mask" behind which other, unexpected properties and functions are hidden. This masquerade has caused great confusion in reproduction research.
One such molecule is a sodium/proton exchanger (NHE) [1], which regulates the pH value in many cells. It belongs to a large family of membrane proteins that transport ions and nutrients into and out of cells - from simple bacteria to humans. Ions pass across the cell membrane in a pas de deux: Flow of sodium ions into the cell is tied to proton efflux (Figure 1). Sodium ions bind to the outside of the exchanger molecule and protons bind to the inside. With to-and-fro movements, as in a rocking-chair, the ions are continuously transported inwards and outwards. This rocking-chair mechanism has been preserved for the transport of molecules as different as ions, amino acids, sugars, or fats. Which mechanisms trigger the rocking movement? It's the ions themselves! When binding to the transport molecule, they simultaneously change its structure: the binding sites move up and down and the ions are carried along as in an elevator. This mechanism is also referred to as alternating access model.
Although the sodium/proton exchanger in sperm had been discovered as early as 2003, its properties were unknown, because the genes could not be functionally expressed in cell lines and the characterization in sperm was technically difficult. We now succeeded to solve thus riddle using the exchanger from sea urchin sperm. The gating mechanism of NHE in sperm is different from that of other exchangers. It carries two other domains known from ion channels: a voltage-sensing domain and a cyclic nucleotide-binding domain (Figure 2). This observation was very surprising and indicated that the additional domains had something to do with the gating. In fact, the sodium/proton exchange is switched on and off by changes in membrane voltage.
The most exciting finding, however, was that we could reveal the role of the cellular messenger cAMP in sea urchin sperm. It has been known for almost 40 years that cAMP is somehow involved in the chemotaxis signalling pathway; yet, the exact function could not be deciphered. Our work shows that cAMP renders the exchanger more sensitive to voltage jumps. This sensitization is reminiscent of pacemaker channels in the heart and the brain [2]. In essence, the exchanger is a chimera between transporters and ion channels. One of the most exciting questions is: How can a voltage-sensor and a cAMP-binding domain control two such different mechanisms of ion transport. The elucidation of the 3-dimensional architecture by biological structural research will hopefully provide the answer as to how the mechanisms of ion transport evolved during evolution.
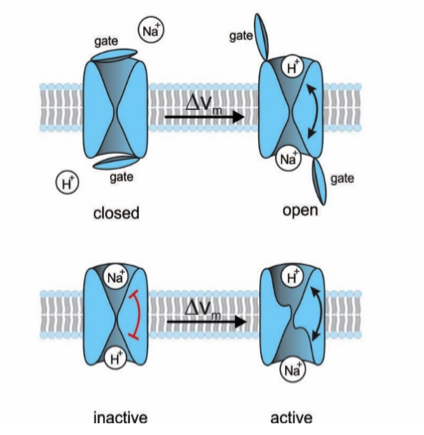
Figure 1: Proposal for two different gating mechanisms of the Na+/H+ exchanger. (top) A lid blocks the ions' access to the respective binding sites. A voltage jump opens the lid; the rocking chair mechanism can now take place. (bottom) Alternatively, there is no lid and the ion-binding sites are accessible, but the rocking-chair movement is locked (red bar). The voltage jump unlocks the mechanism.
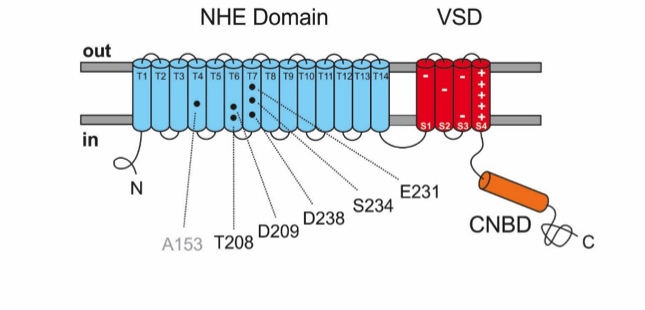
Figure 2: Schematic representation of the NHE topology in the cell membrane. Exchange domain (blue) shows the amino acids (black) of the Na+-binding site, which are conserved between NHEs of archaea and sperm; transmembrane segments are marked as T1-T14. Voltage sensor (red) with positively and negatively charged amino acids; cyclic nucleotide-binding domain (orange).
References
[1] Windler, F., Bönigk, W., Körschen, H.G., Grahn, E., Strünker, T., Seifert, R., Kaupp, U.B. (2018) "The solute carrier SLC9C1 is a Na+/H+-exchanger gated by an S4-type voltage-sensor and cyclic-nucleotide binding" Nature Communications, 9, 2809.
[2] Gauss, R., Seifert, R. & Kaupp, U.B. (1998) "Molecular identification of a hyperpolarization-activated channel in sea urchin sperm" Nature 393, 583-587.